Search Results
223 results found with an empty search
- Omnibus VI Act - New Cosmetics Regulations in the European Union
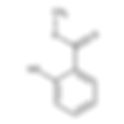
The new regulation known as Omnibus VI will start to apply on December 1, 2023. It is important for cosmetic brands to comply with the Omnibus VI regulations in order to maintain their presence in the European market. This new ingredient ban based on Omnibus Law VI adopted by the European Commission follows a scientific and practical concern regarding chemicals known as CMRs. These substances are characterized by their carcinogenic, mutagenic or reproductive toxicity potential. The legislation covers thirty common ingredients used in cosmetic formulations, which will be explicitly prohibited from inclusion in beauty and personal care products. The main objective of this measure is to comply with Article 15 of the European Union Cosmetics Regulation, which prohibits the presence of CMR substances in cosmetic products. Under this draft, it is established that as of December 1, 2023, cosmetic products containing any of the aforementioned substances may not be available or marketed in the European Union (EU). This action seeks to ensure the protection of public health and consumer safety, as well as to promote the use of safe ingredients in beauty and personal care products. The following substances will be banned for use in cosmetics: ammonium bromide; bis(2-dibutyltin ethylhexanoate); di(dibutyltin acetate); tellurium dioxide; barium diboron tetroxide; 2,2-dimethylpropan-1-ol, derived from tribromo; 3-bromo-2,2-bis(bromomethil)propan-1-ol; 2,4,6-tri-terc-butylphenol; 4,4'-sulfonyldiphenol; bisphenol S; benzophenone; Quinoclamine (ISO); 2-amino-3-chlorine-1,4-naphthoquinone; perfluoroheptanoic acid; tridecafluoroheptanoic acid; N-(isopropoxycarbonyl)-L-valil-(3RS)-3-(4-chlorophenyl)-β-methyl alaninate; valifenalate; 6-[C12-18-alkyl-(branched, unsaturated)-2,5-dioxopyrrolidin-1-il]hexanoic, sodium salts and tris(2-hidroxietil)ammonium acid; 6-[(C10-C13)-alkyl-(branched, unsaturated)-2,5-dioxopyrrolidin-1-il]hexanoic acid; 6-[C12-18-alkyl-(branched, unsaturated)-2,5-dioxopyrrolidin-1-il]hexanoic acid theophylline; 1,3-dimethyl-3,7-di-hydro-1H-purine-2,6-diona; 1,3,5-triazine-2,4,6-triamine; melamine; Fluopicolide (ISO); 2,6-dichlorine-N-[3-chlorine-5-(trifluoromethyl)-2-pyridylmethyl]benzamide; Triamide N-(2-nitrophenyl)phosphoric; N-(5-chlorine-2-isopropylbenzyl)-N-cyclopropyl-3-(difluoromethyl)-5-fluoro-1-methyl-1H-pyrazole-4-carboxamide; isoflucipram; Reaction mass of 3-(difluoromethyl)-1-methyl-N-[(1RS,4SR,9RS)-1,2,3,4-tetrahydro-9-isopropyl-1,4-metanonaftalen-5-il]pyrazole- 4-carboxamide and 3-(difluoromethyl)-1-methyl-N-[(1RS,4SR,9SR)-1,2,3,4-tetrahydro-9-isopropyl-1,4-metanonaftalen-5-il]pyrazole-4-carboxamide [>78% of isomers sin <15% of relative content of anti-isomers]; isopyrazam; Loamy, ext. Of the Azadirachta grains indicates the water extracted and afterwards, they are processed with organic solvents; Cumenian; Diacrylate of 2-ethyl-2-[[(1-oxoallyl)oxy]methyl]-1,3-propanediol; acrylate of 2,2-bis(acryloyloxymethyl)butyl; triacrylate of trimethylolpropane; Pentapotassium 2,2',2'',2''',2''''-(ethane-1,2-diylnitrile)pentaacetate; N-carboxymethyliminobis(ethylene nitrile)tetra(Acetic Acid); (carboxylatemethyl)iminobis(ethylene nitrile)tetraacetate of pentasodium; Acetamiprid (ISO); (1E)-N-[(6-chloropyridin-3-il)methyl]-N'-cyan-N-methylethanimidamide; (E)-N1-[(6-chlorine-3-pyridyl)methyl]-N2-cyan-N1-methylacetamidine; Pendimethalin (ISO); N-(1-ethylpropyl)-2,6-dinitro-3,4-xylidene; Bentazone (ISO); 3-isopropyl-2,1,3-benzothiadiazine-4-one-2,2-dioxide. For more information on Omnibus VI Act, please click here: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32022R0692
- FEI number: Requirement for cosmetics manufacturers
Last December 2022, the US Congress passed the Modernization of the Cosmetic Act, known as MoCRA, which changes the regulatory landscape for cosmetic products in the US. In March 2023, the FDA rescinded all previous registrations of cosmetic products made in the Cosmetic Voluntary Registration Program, making new registrations of all cosmetic products required and mandatory in the new MoCRA portal, available in October 2023. In order to register cosmetic products, companies must indicate the manufacturers FEI number in the registration process. FEI, or FDA Establishment Identifier, is the registration number that a manufacturer obtains when it is registered with the FDA. Until now, FEI registration number was only required for drug, medical device and food manufacturers. From now on, the FEI number is also required for cosmetics manufacturers. To register with the FDA and obtain the FEI number cosmetic manufacturers must have an US Agent I means, a natural or legal person established in the USA who will be in charge of submitting the documentation and will act as a main contact between the FDA and the companies. Each facility where manufacturers carry out manufacturing activities (manufacturing, packaging, storage) must have a different EIF number, in case activities are carried out in different production centers. Products that do not have a registered manufacturer with a valid EIF number cannot be registered in the U.S. market and therefore cannot be sold in the country as of December 29, 2023. Therefore, all cosmetic manufacturing companies have to legalize their status in the U.S. before this date. Belab Services LLC, based in Miami (Florida) provides manufacturers the service of US Agent for registration and obtention of the FEI number of their facilities. For more information, do not hesitate to contact us.
- The draft of the MoCRA Product and Facility Registration has been published
On August 7th, the FDA released the draft of the cosmetic product and facility registration in the United States, in compliance with the new requirements approved in December 2023 under the Modernization of the Cosmetic Act (MoCRA). The new cosmetic product regulations in the USA mandate that anyone who owns or operates a facility engaged in the manufacturing or processing of a cosmetic product for their distribution in the United States must register each facility in the FDA. Additionally, MoCRA mandates the mandatory registration of all cosmetic products through an US Responsible Person. The old (and voluntary) VCRP registration program has been invalidated, rendering all registrations submitted to date null and void. The FDA intends to launch the new electronic submission portal in October 2023. As described in the MoCRA Draft Guidance, the FDA Establishment Identifier (FEI) will be used as the required facility registration number. Therefore, facility owners or operators must obtain an FEI number before submitting the facility registration. Remember that MoCRA has conferred new responsibilities upon the FDA: • Facility Registration: All existing facilities subject to the new FDA MoCRA regulations must register within one year (by December 2023), including contract manufacturers. New facilities must also register within 60 days and renew every two years as required by the FDA. • Product Listing: The Responsible Person must list every cosmetic product marketed with the FDA, including product ingredients, and provide updates annually. Each newly created product now requires the submission of a cosmetic product list detailing everything from ingredients, including fragrance or flavor, to the responsible party for manufacturing facilities and their location. In addition to the mentioned information, the new MoCRA Draft Guidance specifies: The legal requirement to submit cosmetic product facility records and product listings. Who is responsible for making the submissions. What information to include in the submissions. How to make the submissions. When to make the submissions. BELAB SERVICES LLC, located in Miami, Florida, offers its expertise to companies for registering their facilities and products in strict compliance with the new regulations, acting as an US Agents and US Responsible Person for their products and facilities in the USA. For more information, please contact us at: consult@belabservices.com // info@belabservices.com
- Omnibus Act VI approved
The European Commission has officialy approved the Omnibus Act VI, on 20 July, 2023 which add new ingredients to the Annex II of the 1223/2009 European cosmetic regulation. In fact, new 30 cosmetic ingredients will be banned on December 1, 2023. The complete list of new banned ingredients is: According to the Omnibus Act VI draft, the following ingredients will be prohibited for use in cosmetics starting from December 1, 2023: Ammonium bromide; Dibutyltin bis(2-ethylhexanoate); Dibutyltin di(acetate); Tellurium dioxide; Barium diboron tetraoxide; 2,2-dimethylpropan-1-ol,tribromo derivative; 3-bromo-2,2-bis(bromomethyl)propan-1-ol; 2,4,6-tri-tert-butylphenol; 4,4’-sulphonyldiphenol; bisphenol S; Benzophenone; Quinoclamine (ISO); 2-amino-3-chloro-1,4-naphthoquinone; Perfluoroheptanoic acid; tridecafluoroheptanoic acid; methyl N-(isopropoxycarbonyl)-L-valyl-(3RS)-3-(4-chlorophenyl)-β-alaninate; valifenalate; 6-[C12-18-alkyl-(branched, unsaturated)-2,5-dioxopyrrolidin-1-yl]hexanoic acid, sodium and tris(2-hydroxyethyl)ammonium salts; 6-[(C10-C13)-alkyl-(branched, unsaturated)-2,5-dioxopyrrolidin-1-yl]hexanoic acid; 6-[C12-18-alkyl-(branched, unsaturated)-2,5-dioxopyrrolidin-1-yl]hexanoic acid; Theophylline; 1,3-dimethyl-3,7-dihydro-1H-purine-2,6-dione; 1,3,5-triazine-2,4,6-triamine; melamine; Fluopicolide (ISO); 2,6-dichloro-N-[3-chloro-5-(trifluoromethyl)-2-pyridylmethyl]benzamide; N-(2-nitrophenyl)phosphoric triamide; N-(5-chloro-2-isopropylbenzyl)-N-cyclopropyl-3-(difluoromethyl)-5-fluoro-1-methyl-1H-pyrazole-4-carboxamide; isoflucypram; Reaction mass of 3-(difluoromethyl)-1-methyl-N-[(1RS,4SR,9RS)-1,2,3,4-tetrahydro-9-isopropyl-1,4-methanonaphthalen-5-yl]pyrazole-4-carboxamide and 3-(difluoromethyl)-1-methyl-N-[(1RS,4SR,9SR)-1,2,3,4-tetrahydro-9-isopropyl-1,4-methanonaphthalen-5-yl]pyrazole-4-carboxamide [>78% syn isomers <15% anti isomers relative content]; isopyrazam; Margosa, ext. from the kernels of Azadirachta indica extracted with water and further processed with organic solvents; Cumene; 2-ethyl-2-[[(1-oxoallyl)oxy]methyl]-1,3-propanediyl diacrylate; 2,2-bis(acryloyloxymethyl)butyl acrylate; trimethylolpropane triacrylate; Pentapotassium 2,2’,2’’,2’’’,2’’’’-(ethane-1,2-diylnitrilo)pentaacetate; N-carboxymethyliminobis(ethylenenitrilo)tetra(acetic acid); Pentasodium (carboxylatomethyl)iminobis(ethylenenitrilo)tetraacetate; Acetamiprid (ISO); (1E)-N-[(6-chloropyridin-3-yl)methyl]-N’-cyano-N-methylethanimidamide; (E)-N1-[(6-chloro-3-pyridyl)methyl]-N2-cyano-N1-methylacetamidine; Pendimethalin (ISO); N-(1-ethylpropyl)-2,6-dinitro-3,4-xylidene; Bentazone (ISO); 3-isopropyl-2,1,3-benzothiadiazine-4-one-2,2-dioxide. Times: products with these ingredients cannot be placed on the market from this data. If you need assistance please, contact us.
- MD IN THE UK! UK GOVERMENT ANNOUNCES THE EXTENSION OF CE (EUROPE) MARK RECOGNITION WITH NO DEADLINE
The Department for Business and Trade (DBT) has announced its intention to indefinitely extend the use of CE marking for businesses. Following extensive engagement with industry, British firms will be able to continue the use of CE marking alongside UKCA, invalidating the last deadline of December 2024. Due to this fact, CE marked Medical Devices can be placed on the UK market without specifically obtaining the UKCA mark even though UKCA mark will be optional for these CE Medical Devices. However, we have to remember that the obligation of MD manufacturers to register their products in the MHRA portal, having an UK Responsible Person, still being in force. For products that are not CE marked, the UKCA mark remains mandatory. BELAB SERVICES helps your company to comply with UK Medical Devices regulations, ensuring product compliance, elaborating the Technical File, making the MHRA MD registration and acting as an UK Responisble Person/ UK Authorized Representative.
- Attention! MoCRA - VCRP has been STOPPED!
The U.S. Food and Drug Administration (FDA) has stopped accepting submissions to the Voluntary Cosmetic Registration Program (VCRP) effective March 27, 2023 as a result of FDA’s plans to develop a program for submission of the facility registrations and product listings mandated by the “Modernization of Cosmetics Regulation Act of 2022” (MoCRA). The new registration program is not still developed so from now (and until new program launch) no more cosmetic products notifications are possible. MoCRA will change the rules of cosmetic products in the USA. What are the new 2024 changes? - All manufacturers and packers shall register their facilities under a US Agent responsibility. - Al products shall have a "Responsible Person" in the US, it means, a US entity which appears on the product labels and act as regulatory a contact between FDA and the brand. - Mandatory registration of each product in the new registration program. - Possible changes in ingredients allowance and label requirements. To manage all this changes, BELAB SERVICES LLC offers all this services by our new Miami (FL) subsidiary. If you need more information please, do not hesitate to contact us. consult@belabservices.com
- Final SCCS Opinion on Alpha and Beta Arbutin
The Scientific Committee on Consumer Safety (SCCS) published its final opinion on alpha and beta arbutin on 1 February 2023. The final opinion has a positive conclusion, compared to its initial assessment in the draft opinion. The SCCS concluded that: alpha-arbutin is safe when used in face creams up to a maximum concentration of 2% and in body lotions up to a concentration of 0.5%, also when used together; beta-arbutin is safe when used in face creams up to a maximum concentration of 7%; aggregate exposure of alpha-arbutin (2% in face cream and 0.5% in body lotion) with beta-arbutin (7% in face cream) is also considered safe. Furthermore, hydroquinone should remain as low as possible in formulations containing alpha-or beta-arbutin and should not be higher than the unavoidable traces in both arbutins. In the new studies, submitted by the applicant, 3ppm was the limit of quantification for hydroquinone and 1ppm for the limit of the detection. If you need regulatory assistance, please, do not hesitate to contact us. consult@belabservices.com
- MoCRA - How affects the Modernization of Cosmetics Regulation Act
Last December, the US Congress passed The Modernization of Cosmetics Regulation Act of 2022 (MoCRA), which creates federal standards for cosmetic cosmetic safety and compliance. How cosmetic companies around the World will be affected? Here a list of the main points to considerate to be in compliance with new regulations: 1. Registration and Product Listings Cosmetic companies have to to register their facilities and submit product listings to FDA. It also requires the Responsible Person (i.e., a “manufacturer, packer, or distributor of a cosmetic product whose name appears on the label of such cosmetic product) to submit to FDA the cosmetic product’s listing, including the ingredients of any fragrances or flavours. The registration shall be done no later than one year after the enactment date. Companies must renew a product listing annually with any updates. 2. Safety Substantiation Requires manufacturers to maintain records demonstrating adequate substantiation of the safety of a cosmetic product, such as reputable “tests or studies, research, analyses, or other evidence or information… sufficient to support a reasonable certainty that a cosmetic product is safe.” It means that Cosmetic Product Safety Report shall be done before the product marketing. 3. Good Manufacturing Practice FDA is going to promulgate good manufacturing practice (GMP) regulations for cosmetics manufacturing and processing facilities with the intention of protecting the public health and ensuring that cosmetic products distributed in the United States are not adulterated. In developing this guidance, FDA incorporated elements from the International Organization for Standardization’s standard for cosmetic GMPs (ISO 22716:2007). It may therefore be useful to track any developments in ISO 22716:2007 going forward because these will likely form the basis of any GMPs promulgated by FDA. 4. Adverse Event Reporting The Responsible Person shall maintain records of health-related adverse events associated with the use of its product for six years (or three years for small businesses), and to report to FDA any serious adverse events within 15 business days of learning about the event. 5. Recalls FDA has been authorized for requesting a voluntary recall of a cosmetic product if the agency determines that there is a reasonable probability that the product is adulterated or misbranded. If the responsible person does not comply with that request, FDA may order a mandatory recall (subject to requirements for an informal hearing). It means that the Responsible Person has the opportunity to recall the product, but if refused, FDA, by order, can call for the immediate cease distribution and recall the product. Considerations which may help cosmetics companies may include: Consider to hire a specialized professional to ensure full compliance with new standards; Careful creation of not only processes and procedures for implementing all MoCRA requirements but also training of employees on the importance of compliance and the dangers of non-compliance, as well as careful documentation practices; Timely and complete registration and product listing disclosures to FDA; Review of safety substantiation tests and data sources for each product to ensure they are scientifically supported and can be defended, including the test methods utilized themselves in addition to their results; Updating or establishing new Standard Operating Procedures to ensure compliance with forthcoming GMPs.
- Recuerda! Nuevas Restricciones al Methyl Salicilate en cosméticos
On May 28, the Official Regulation (EU) 2021/849 was published which modifies the classification of Methyl Salicylate as a CMR substance (Repr.2). Due to this reason, the use of Methyl Salicylate has been restricted again in Annex III. The entry into force of the new restrictions will be on December 17, 2022. From this date, products that do not comply with the new conditions of use cannot be placed on the market or marketed. THERE WILL BE NO TRANSITIONAL PERIODS NUEVAS CONDICIONES DE USO DEL SALICILATO DE METILO: In addition, its use in products for children under 6 years of age is prohibited, except in toothpastes whose maximum concentration of use will be 2.52%. If you are affected with this new restriction please, do not hesitate to contact us. consult@belabservices.com
- Cosmetics in the USA: FDA Compliance
Increasingly companies decide to export their cosmetics and the United States is a very attractive market with more than 300 million consumers. Nevertheless, Federal Drug Administration (FDA) rules are one of the most restrictive and complex of the world. Also, we have to take into account State laws and regulations across the nation, as California 65 Proposition. To export cosmetics with success it is mandatory to analyse and adapt cosmetics products under FDA compliance specialist. Formula, labels and claims are one of the key factors to check and correct to avoid export and customs problems. If you want to leave it in the capable hands Belab Services Consulting can help you to have FDA compliance cosmetics and OTC products. For more information, contact us and we will delight to help you. consult@belabservices.com www.belabservices.com
- Italian Law on Packaging Waste Labeling
The Italian Ministry of Ecological Transition (Ministero della transizione ecológica) has published the document “Labelling for packaging -Guidelines according to Art. 219 paragraph 5 of Legislative Decree 152/2006 and subsequent modifications and integrations”, for the correct management of packaging waste, also known as “Italian Law on Packaging Waste Labeling”. The document explains the general rules for making a compliance packaging according to the law and how to place the information on it. These new labelling requirements come into force on 1th January 2023 for products placed in the Italian market. One of the novelties is the obligation to identify manually separable parts of the products packaging and also to identify the packaging materials with the ISO 1043-1 (ISO 10667-1 for recycled polymers) codes. In addition, companies shall to place on the labels the consumer instructions for waste collection following the international colour codes for the different kinds of waste. One of the most important things is that the law allows the inclusion of this information through digital channels (QR, websites) in case of small packaging (ones with surface less than 25cm2 or packaging with capacity up to 125ml). This Guideline encourages companies to add more information as mandatory. For example, companies can: - Identify the manually separable components of the packaging by name (CAP, BOTTLE, CASE, FLACON, JAR...). - Introduce additional waste management instructions. For example: Check your local municipal Guidelines. Only dispose of empty containers Empty the packaging of its content before recycling Separate collection Reduce the volume of the can / box Separate the label and dispose of it correctly Remove the cap from the container The guidance includes some visual illustrations as follows: Belab Services supports the industry in the compliance of the new Italian regulatory requirements. If your company is affected by the new regulations and you need help or more information please, do not hesitate to contact us!
- TRIMAN symbol and how it applies to cosmetic products
The new AGEC law against waste and for the Circular Economy requires the TRIMAN marking of cosmetic products. As of January 1, 2022, the new French AGEC law stipulates the obligation to mark all products put up for sale on the French market with the TRIMAN symbology. The TRIMAN symbol indicates that a packaging is recyclable and helps consumers to correctly manage waste. To correctly mark our products we can follow these simple rules: TRIMAN logo + Tri Facile text (optional) + product components + container symbol. The following examples applied to cosmetics can be very useful: a. Tube: toothpaste, cream, facial mask, foundation, etc. b. Fragrance: As you can see, there is not only one way to make the TRIMAN logo, but we have several options (with more text, without text, with symbols...). The important thing is to do it correctly and that the information is available. If your company needs help to apply the TRIMAN symbology and comply with the requirements of the AGEC law, do not hesitate to contact us! consult@belabservices.com